Cancer research is changing fast. For decades, scientists studied cancer cells on flat plastic dishes, a method that never truly showed how real tumors behave inside the human body. Today, 3D tumoroid models are turning that old approach upside down. These tiny, lab-grown, three-dimensional clusters of cancer cells are giving doctors a much clearer picture of how cancer works. As a result, finding better treatments is faster, safer, and more personal than ever before.
What Are 3D Tumoroid Models and Why Do They Matter?
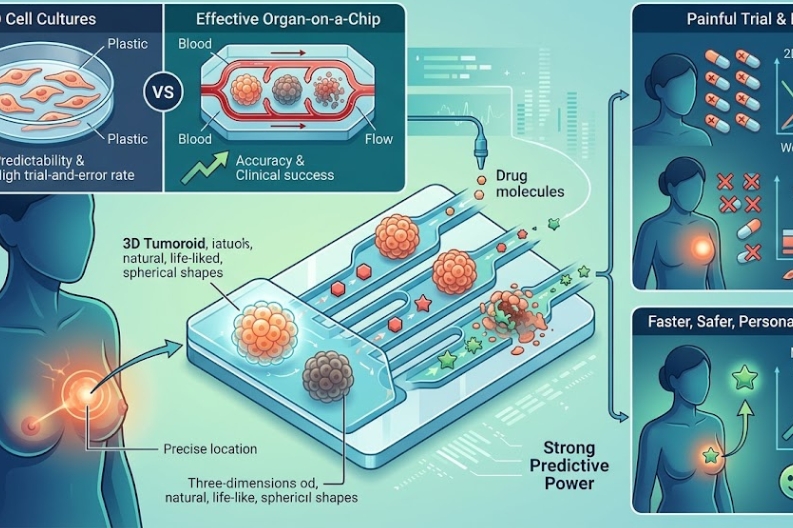
A tumoroid is a miniature, simplified version of a real patient’s tumor, grown in a laboratory dish. Unlike flat 2D cell cultures, tumoroids grow in a natural spherical shape. This 3D structure closely mirrors how actual tumors develop inside the human body.
Because of this shape, the outer cells receive oxygen while the inner cells struggle for nutrients, just like in a real tumor. This is critical for researchers. It allows them to see precisely how a drug penetrates a solid tumor mass. Therefore, doctors and biotech companies can predict whether a treatment will work, before it ever reaches a patient.
How Tumoroids Mimic Real Tumors in the Human Body?
In the body, cells constantly communicate with each other. These 3D models support that “cell talk,” making research far more realistic and reliable. Traditional flat cell cultures simply cannot replicate this. That is why results from 2D studies often do not translate into real clinical success.
Why Old Cancer Research Methods Are No Longer Enough?
Modern medicine is moving toward a fully personalized approach. However, old research methods have major limitations. Here is a quick comparison:
Comparing 2D Cell Culture, Animal Testing, and 3D Tumoroids
| Feature | 2D Cell Culture | Animal Testing | 3D Tumoroid Models |
| Cell Shape | Flat / Distorted | Complex | Natural / Spherical |
| Human DNA | Often yes | No | Always yes |
| Cost | Low | Very high | Moderate |
| Predictive Power | Weak | Variable | Very strong |
| Animal Use | None | High | None |
Clearly, tumoroids offer a much better balance of accuracy, ethics, and cost. Furthermore, they use human cells, which means the data is directly relevant to human patients.
How 3D Tumoroid Models Help Find the Best Cancer Drug for You?
One of the most exciting applications of this technology is personalized cancer treatment. Scientists grow a patient’s specific tumor cells in a lab. They then test multiple drugs on those tumoroids to see which one works best. This is precision medicine in action.
Real-Life Example: Breast Cancer and Personalized Treatment
Consider a breast cancer patient. Doctors take a small tissue sample from her tumor. In the lab, they grow her exact tumor cells as 3D tumoroids. Next, they test ten different chemotherapy drugs on these models. The drug that most effectively destroys the tumoroid is then chosen for her treatment.
This completely removes the painful “trial and error” process. Consequently, patients get faster relief, fewer side effects, and a much higher chance of recovery. According to the National Cancer Institute, this kind of targeted research is one of the most promising frontiers in oncology today.
Organ-on-a-Chip: The Next Step in Biotech Innovation
The biotech world is taking these models even further. Scientists are now placing 3D tumoroids onto tiny microchips that simulate blood flow, a technology known as Organ-on-a-Chip. This allows researchers to watch in real time how a drug travels through veins and reaches a tumor.
This level of detail was simply impossible just a few years ago. The Wyss Institute at Harvard is leading some of the most advanced work in this area. Their research shows that this technology could soon replace many types of early-stage clinical drug trials entirely.
Additionally, this innovation is helping scientists study conditions beyond cancer – including Alzheimer’s disease, heart disease, and diabetes. As a result, the benefits extend well beyond oncology.
Current Challenges and What Scientists Are Working On
Despite the huge promise, a few challenges still remain. Here is what researchers are actively working to solve:
- Cost and equipment – Growing high-quality tumoroids still requires specialized lab tools.
- Adding blood vessels – Real tumors are surrounded by blood vessels and immune cells. Scientists are now working to include these in the models.
- Standardization – The National Cancer Institute is actively funding efforts to ensure every lab worldwide produces consistent, reliable results.
- Scale-up – Testing thousands of drug combinations at once requires advanced automation technology.
Nevertheless, progress is rapid. Each year, these models become more detailed and more life-like. Scientists are confident that most of these challenges will be solved within the next decade.
The Future of Cancer Treatment and Long-Term Health
The shift toward 3D tumoroid models is not just a trend, it is a fundamental change in how we understand and fight cancer. By using these lab-grown tumors, doctors can move away from guesswork and toward truly personalized care. Patients are no longer treated as statistics. They are treated as individuals with unique biology.
Moreover, the knowledge gained from tumoroids is also informing research into nutrition, supplements, and healthy lifestyle habits. Understanding how cells behave in a 3D environment helps scientists give better, evidence-based advice on diet and long-term wellness. This is why many experts believe that 3D tumoroid models will soon become a standard tool in clinics worldwide, not just research labs. The future of cancer treatment is here, and it is personal.